Uncategorized
J&J CEO announces a lead COVID-19 vaccine candidate
J&J CEO announces a lead COVID-19 vaccine candidate


CNBC transcript: Johnson & Johnson CEO Alex Gorsky Speaks with CNBC’s “Squawk Box” today and announces that it’s now selected a lead COVID-19 vaccine candidate
WHEN: Today, Monday, March 30, 2020
WHERE: CNBC’s “Squawk Box”
Q4 2019 hedge fund letters, conferences and more
Johnson & Johnson CEO On How Close Company Is To COVID-19 vaccine
ANDREW ROSS SORKIN: Welcome back to "Squawk Box" this morning. We have some big news on the fight against the coronavirus. Last hour Johnson & Johnson announcing it’s now selected a lead COVID-19 vaccine candidate and says it expects to begin Phase 1 human clinical trials by September, at the latest. Joining us now is Johnson & Johnson CEO Alex Gorsky. And we’re thrilled to have him on the program. Alex, thank you for joining us. This is very good news. Tell us about what’s gone into this and why you are so optimistic about it.
ALEX GORSKY: Well, Andrew, and Joe, and Becky, thank you very much for having me on, though it’s always tough to follow Senator Cruz. But, you know, we’re very excited about the announcement that we’re making this morning about the partnership we’re doing with BARTA, which is the government agency responsible for pandemics and vaccines and what we’re doing to accelerate our development but also, our production for something against COVID-19.
And you know, really, this started decades ago. We’ve been working in the immunology field for some time. We’ve been working in areas, for example, like SARS, Ebola, HIV. And I think what gives us confidence is based upon the work that we have done with this COVID-19 vaccine platform, we have very good early indicators that not only can we depend on this to be a safe COVID-19 vaccine base, but also one that will ultimately be effective based on all the early testing and modeling that we’ve been doing
You know, as you mentioned earlier, we expect to begin first human testing in September. But, in parallel, not only do you need a safe and effective vaccine but you also need to have one that can be produced in large volumes. We are going to be doing that at risk simultaneously right here in the United States. And we expect to have results, interim results at least, from our trials likely in December at the latest early January.
That should put us in position early in 2021 to literally have hundreds of millions of doses available and by the end of the year up to 1 billion. That’s our plan. That’s the reason why we’re confident. We understand we have got more testing. We still have a lot more work to do. But this is a bit of a moonshot for J & J, but going forward, it’s one we feel is very important for us to be doing at this point in time.
ANDREW ROSS SORKIN: So, Alex, in terms, though, of the timeline, the ability to get this into the marketplace, if you will, you’re talking about early 2021 for lots of people to get this. Is that just for special access? Or are you talking really about next summer? What’s that timeline looking like in your mind?
ALEX GORSKY: Sure, Andrew. I don’t mean to sound like an economist, it is going to depend on a few factors. But let me tell you why we’re optimistic. If we have the interim results from clinical trials late this year and let’s just project forward. A time where frankly there still is significant concern about the pandemic as we head into 2021, we will have the ability to work with regulators, to determine, first of all, that we have a safe and effective COVID-19 vaccine based on trial results within the first quarter of next year.
And because we will have made the efforts to open up a production facility here in the U.S., to do all the technology transfer that is required, we should also be in a position to have a very significant number of dosages available in that Q1 Q2 period. Now, look, we are going to l have to learn more.
We’ll have to see exactly what our yield is, but we’re very confident in the underlying technology that we have, that we -- that we can have that level of dose ages available and ultimately make the right decision early in 2021 about, you know, what’s in the best public health interest and how we proceed.
ANDREW ROSS SORKIN: Alex, what is this coronavirus vaccine going to cost the consumer, do you imagine? And your stock has moved on this number. Is this going to be profitable for you?
ALEX GORSKY: Well, look, this is about doing right but for global public health. And as we said in our statement, we want our number one concern after bringing it out and demonstrating that it’s safe and effective is to make sure that it’s accessible and affordable on a global level. In fact, we’re taking this step further and we’ll do this on a not for profit basis. We’re working jointly with the government on this where we’ll be putting more than $1 billion worth of investment in terms of the research and development, but also scaling up the production capacity on this. You know, in the very near-term.
And so that is our expectation it’s difficult for me to say an exact price because we still have to determine some of these other factors be. We want to make sure that everyone can get access and that we can do so consistent with what you see other commonly used COVID-19 vaccines for here in our country and around the world.
JOE KERNEN: Hey Alex, you had your economist hat on for a second. Put on your scientist hat for me. Tell me what you’re keying on in terms of basing the coronavirus vaccine on. Is it something on the surface of the virus? Is something that is in the nucleic acids, the RNA itself? And is it -- why are you confident this is conserved? In other words, if there’s mutations, this will be essential to the way the virus works, so it will continue to work, even if there is a mutation.
ALEX GORSKY: Joe, appreciate the question. And let me respond this way. First of all, we have a lot of confidence. Because, you know, when you’re building a vaccine, you’ve got to think about it -- one way to think about it is like a car. And the body of the car, the platform that we’re using is one we have already demonstrated safety and efficacy in things like SARS, which you know as a biologist, is a similar condition or similar science to what we’re seeing with COVID-19. We’ve also used it in older patients for Ebola, for example, in younger patients. And we have seen the safety to be, you know, very consistent.
The next step, of course, is okay, what kind of a motor, what kind of software can we insert into that car that will be effective against COVID-19? What we know about all of the work we’ve done in coronavirus vaccines is the early testing that we’re doing, both in Vivo and in some animal models, tends to be predictive of ultimately what you’re going to see in the human trials itself.
As you know, coronavirus, it’s a tough coronavirus. It’s an RNA virus. So, we’re -- based upon what we’ve seen so far, we think we have the right approach. Obviously, we wouldn’t be taking these other steps if we didn’t feel that way. But we do have to conduct the human trials and we’ll learn more through that process as we go through the fourth quarter of this year.
JOE KERNEN: You can scale up manufacturing quickly and get a lot of it. It’s not like some of the vaccines that are very difficult to make.
ALEX GORSKY: Joe, that’s a great point. And as I’m sure you’re aware, with traditional vaccines, frequently you’re actually working with an egg, like a chicken egg, where you may be able to yield a couple of doses per egg. It has to be done on the surface of a very large vat, and that of course takes time and can result in years of development. What is unique about our approach -- and it’s interesting because actually from a technology, another COVID-19 vaccine platform that we’re going after, initially we didn’t achieve some of our objectives, that frequently happens in investment, and research and development. But the production capability, it’s called PER. C6, and basically what that does, instead of having to use an egg or instead of having to have it on the surface, this can be done where it’s very -- in an intense environment.
So, think of a thousand-liter beaker, where these proteins can be harvested. They don’t need to be on the surface. And so, we can get high yield. So, for example, in a 1,000-liter beaker, we’re getting yields of hundreds of millions of coronavirus vaccines. And that, of course, is what gives us the confidence that not only will we have a vaccine that works but we can produce it in the kinds of volumes that are really going make the difference and frankly tamp down the curve and, you know, eventually prevent this virus from happening in the first place.
BECKY QUICK: Alex, that’s kind of related to my question. I know you that guys are taking what would normally take five to six years and compressing it into five to six months. I’m just wondering, is that because technology is advancing so quickly? Is that because you’re pouring a billion dollars’ worth of resources into it or because the FDA is relaxing some of its guidelines? What gets you to that point?
ALEX GORSKY: You know, Becky, it’s all of the above it starts with -- huge thank you to Dr. Paul Stoffels, Dr. Johan Van Hoof, people who have poured, you know, a good chunk of their careers into understanding infectious disease and conditions like this. They have literally been working 24/7 to identify, you know, how do we, first of all, put this through the most rigorous tests that are going to give us the signal that indicates it.
And we quickly went through our library, and that’s where now we have focused on this one particular vector that we’re highly confident in. Simultaneously, we’ve been working on all the technology transfer, the production ramp-up. So, another shout out to our global supply chain colleagues who have been working around-the-clock to say how can we do this and how can we do this in the United States, even though that’s not where the original technology was developed? How can we bring it to a level that we can scale up, not compromise at all in regarding safety and quality but do it in the right way? So, all those factors.
Then, of course, the partnership with the United States and I want to thank the government, BARTA, the work they’ve been doing, HHS, also the FDA, they work closely with us. If you do an interim analysis statistically, how can we accelerate that? Again, making sure we’re getting all the information that we need, but also recognize frankly the situation that we’re in around the world.
ANDREW ROSS SORKIN: Alex, we want to thank you for joining us this morning. We want to wish you well. Your success will be the world’s success, and we appreciate you joining us this morning.
ALEX GORSKY: Andrew, Joe, thank you. Look, biology started this, biology and pharmacology will be a big part of winning this in the end, and we are absolutely committed to making that happen. Thank you, guys.
ANDREW ROSS SORKIN: Okay. Alex Gorsky from J&J. We appreciate it. We’ll be back tomorrow. CNBC’s special coverage continues right now.
The post J&J CEO announces a lead COVID-19 vaccine candidate appeared first on ValueWalk.
Uncategorized
February Employment Situation
By Paul Gomme and Peter Rupert The establishment data from the BLS showed a 275,000 increase in payroll employment for February, outpacing the 230,000…
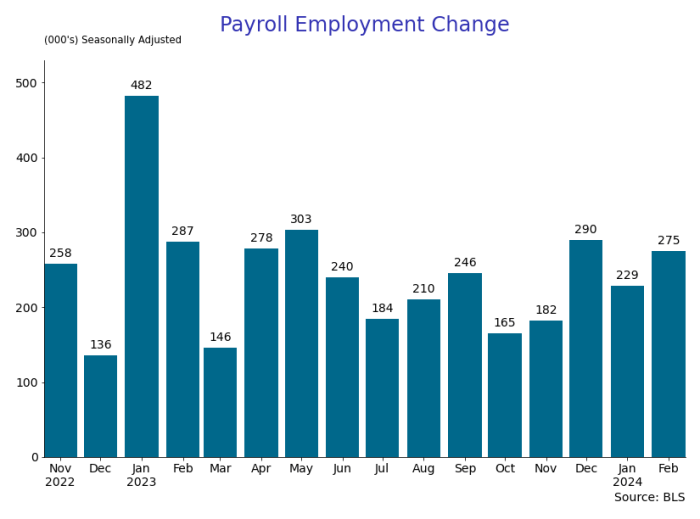
By Paul Gomme and Peter Rupert
The establishment data from the BLS showed a 275,000 increase in payroll employment for February, outpacing the 230,000 average over the previous 12 months. The payroll data for January and December were revised down by a total of 167,000. The private sector added 223,000 new jobs, the largest gain since May of last year.

Temporary help services employment continues a steep decline after a sharp post-pandemic rise.


Average hours of work increased from 34.2 to 34.3. The increase, along with the 223,000 private employment increase led to a hefty increase in total hours of 5.6% at an annualized rate, also the largest increase since May of last year.

The establishment report, once again, beat “expectations;” the WSJ survey of economists was 198,000. Other than the downward revisions, mentioned above, another bit of negative news was a smallish increase in wage growth, from $34.52 to $34.57.

The household survey shows that the labor force increased 150,000, a drop in employment of 184,000 and an increase in the number of unemployed persons of 334,000. The labor force participation rate held steady at 62.5, the employment to population ratio decreased from 60.2 to 60.1 and the unemployment rate increased from 3.66 to 3.86. Remember that the unemployment rate is the number of unemployed relative to the labor force (the number employed plus the number unemployed). Consequently, the unemployment rate can go up if the number of unemployed rises holding fixed the labor force, or if the labor force shrinks holding the number unemployed unchanged. An increase in the unemployment rate is not necessarily a bad thing: it may reflect a strong labor market drawing “marginally attached” individuals from outside the labor force. Indeed, there was a 96,000 decline in those workers.


Earlier in the week, the BLS announced JOLTS (Job Openings and Labor Turnover Survey) data for January. There isn’t much to report here as the job openings changed little at 8.9 million, the number of hires and total separations were little changed at 5.7 million and 5.3 million, respectively.

As has been the case for the last couple of years, the number of job openings remains higher than the number of unemployed persons.

Also earlier in the week the BLS announced that productivity increased 3.2% in the 4th quarter with output rising 3.5% and hours of work rising 0.3%.

The bottom line is that the labor market continues its surprisingly (to some) strong performance, once again proving stronger than many had expected. This strength makes it difficult to justify any interest rate cuts soon, particularly given the recent inflation spike.
unemployment pandemic unemploymentUncategorized
Mortgage rates fall as labor market normalizes
Jobless claims show an expanding economy. We will only be in a recession once jobless claims exceed 323,000 on a four-week moving average.
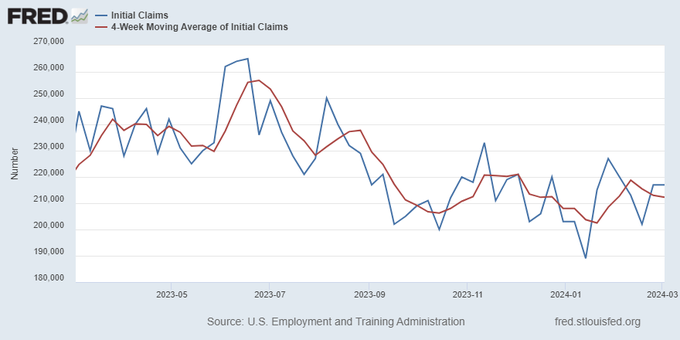
Everyone was waiting to see if this week’s jobs report would send mortgage rates higher, which is what happened last month. Instead, the 10-year yield had a muted response after the headline number beat estimates, but we have negative job revisions from previous months. The Federal Reserve’s fear of wage growth spiraling out of control hasn’t materialized for over two years now and the unemployment rate ticked up to 3.9%. For now, we can say the labor market isn’t tight anymore, but it’s also not breaking.
The key labor data line in this expansion is the weekly jobless claims report. Jobless claims show an expanding economy that has not lost jobs yet. We will only be in a recession once jobless claims exceed 323,000 on a four-week moving average.
From the Fed: In the week ended March 2, initial claims for unemployment insurance benefits were flat, at 217,000. The four-week moving average declined slightly by 750, to 212,250
Below is an explanation of how we got here with the labor market, which all started during COVID-19.
1. I wrote the COVID-19 recovery model on April 7, 2020, and retired it on Dec. 9, 2020. By that time, the upfront recovery phase was done, and I needed to model out when we would get the jobs lost back.
2. Early in the labor market recovery, when we saw weaker job reports, I doubled and tripled down on my assertion that job openings would get to 10 million in this recovery. Job openings rose as high as to 12 million and are currently over 9 million. Even with the massive miss on a job report in May 2021, I didn’t waver.
Currently, the jobs openings, quit percentage and hires data are below pre-COVID-19 levels, which means the labor market isn’t as tight as it once was, and this is why the employment cost index has been slowing data to move along the quits percentage.

3. I wrote that we should get back all the jobs lost to COVID-19 by September of 2022. At the time this would be a speedy labor market recovery, and it happened on schedule, too
Total employment data
4. This is the key one for right now: If COVID-19 hadn’t happened, we would have between 157 million and 159 million jobs today, which would have been in line with the job growth rate in February 2020. Today, we are at 157,808,000. This is important because job growth should be cooling down now. We are more in line with where the labor market should be when averaging 140K-165K monthly. So for now, the fact that we aren’t trending between 140K-165K means we still have a bit more recovery kick left before we get down to those levels.

From BLS: Total nonfarm payroll employment rose by 275,000 in February, and the unemployment rate increased to 3.9 percent, the U.S. Bureau of Labor Statistics reported today. Job gains occurred in health care, in government, in food services and drinking places, in social assistance, and in transportation and warehousing.
Here are the jobs that were created and lost in the previous month:

In this jobs report, the unemployment rate for education levels looks like this:
- Less than a high school diploma: 6.1%
- High school graduate and no college: 4.2%
- Some college or associate degree: 3.1%
- Bachelor’s degree or higher: 2.2%

Today’s report has continued the trend of the labor data beating my expectations, only because I am looking for the jobs data to slow down to a level of 140K-165K, which hasn’t happened yet. I wouldn’t categorize the labor market as being tight anymore because of the quits ratio and the hires data in the job openings report. This also shows itself in the employment cost index as well. These are key data lines for the Fed and the reason we are going to see three rate cuts this year.
recession unemployment covid-19 fed federal reserve mortgage rates recession recovery unemploymentUncategorized
Inside The Most Ridiculous Jobs Report In History: Record 1.2 Million Immigrant Jobs Added In One Month
Inside The Most Ridiculous Jobs Report In History: Record 1.2 Million Immigrant Jobs Added In One Month
Last month we though that the January…
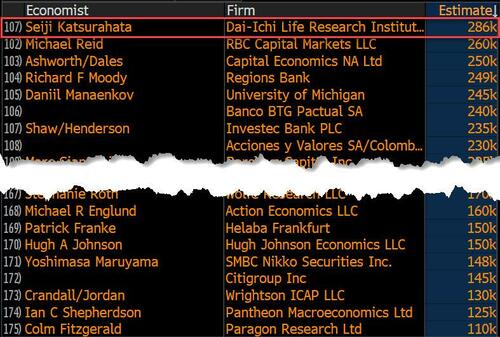
Last month we though that the January jobs report was the "most ridiculous in recent history" but, boy, were we wrong because this morning the Biden department of goalseeked propaganda (aka BLS) published the February jobs report, and holy crap was that something else. Even Goebbels would blush.
What happened? Let's take a closer look.
On the surface, it was (almost) another blockbuster jobs report, certainly one which nobody expected, or rather just one bank out of 76 expected. Starting at the top, the BLS reported that in February the US unexpectedly added 275K jobs, with just one research analyst (from Dai-Ichi Research) expecting a higher number.
Some context: after last month's record 4-sigma beat, today's print was "only" 3 sigma higher than estimates. Needless to say, two multiple sigma beats in a row used to only happen in the USSR... and now in the US, apparently.
Before we go any further, a quick note on what last month we said was "the most ridiculous jobs report in recent history": it appears the BLS read our comments and decided to stop beclowing itself. It did that by slashing last month's ridiculous print by over a third, and revising what was originally reported as a massive 353K beat to just 229K, a 124K revision, which was the biggest one-month negative revision in two years!
Of course, that does not mean that this month's jobs print won't be revised lower: it will be, and not just that month but every other month until the November election because that's the only tool left in the Biden admin's box: pretend the economic and jobs are strong, then revise them sharply lower the next month, something we pointed out first last summer and which has not failed to disappoint once.
In the past month the Biden department of goalseeking stuff higher before revising it lower, has revised the following data sharply lower:
— zerohedge (@zerohedge) August 30, 2023
- Jobs
- JOLTS
- New Home sales
- Housing Starts and Permits
- Industrial Production
- PCE and core PCE
To be fair, not every aspect of the jobs report was stellar (after all, the BLS had to give it some vague credibility). Take the unemployment rate, after flatlining between 3.4% and 3.8% for two years - and thus denying expectations from Sahm's Rule that a recession may have already started - in February the unemployment rate unexpectedly jumped to 3.9%, the highest since February 2022 (with Black unemployment spiking by 0.3% to 5.6%, an indicator which the Biden admin will quickly slam as widespread economic racism or something).
And then there were average hourly earnings, which after surging 0.6% MoM in January (since revised to 0.5%) and spooking markets that wage growth is so hot, the Fed will have no choice but to delay cuts, in February the number tumbled to just 0.1%, the lowest in two years...
... for one simple reason: last month's average wage surge had nothing to do with actual wages, and everything to do with the BLS estimate of hours worked (which is the denominator in the average wage calculation) which last month tumbled to just 34.1 (we were led to believe) the lowest since the covid pandemic...
... but has since been revised higher while the February print rose even more, to 34.3, hence why the latest average wage data was once again a product not of wages going up, but of how long Americans worked in any weekly period, in this case higher from 34.1 to 34.3, an increase which has a major impact on the average calculation.
While the above data points were examples of some latent weakness in the latest report, perhaps meant to give it a sheen of veracity, it was everything else in the report that was a problem starting with the BLS's latest choice of seasonal adjustments (after last month's wholesale revision), which have gone from merely laughable to full clownshow, as the following comparison between the monthly change in BLS and ADP payrolls shows. The trend is clear: the Biden admin numbers are now clearly rising even as the impartial ADP (which directly logs employment numbers at the company level and is far more accurate), shows an accelerating slowdown.
But it's more than just the Biden admin hanging its "success" on seasonal adjustments: when one digs deeper inside the jobs report, all sorts of ugly things emerge... such as the growing unprecedented divergence between the Establishment (payrolls) survey and much more accurate Household (actual employment) survey. To wit, while in January the BLS claims 275K payrolls were added, the Household survey found that the number of actually employed workers dropped for the third straight month (and 4 in the past 5), this time by 184K (from 161.152K to 160.968K).
This means that while the Payrolls series hits new all time highs every month since December 2020 (when according to the BLS the US had its last month of payrolls losses), the level of Employment has not budged in the past year. Worse, as shown in the chart below, such a gaping divergence has opened between the two series in the past 4 years, that the number of Employed workers would need to soar by 9 million (!) to catch up to what Payrolls claims is the employment situation.
There's more: shifting from a quantitative to a qualitative assessment, reveals just how ugly the composition of "new jobs" has been. Consider this: the BLS reports that in February 2024, the US had 132.9 million full-time jobs and 27.9 million part-time jobs. Well, that's great... until you look back one year and find that in February 2023 the US had 133.2 million full-time jobs, or more than it does one year later! And yes, all the job growth since then has been in part-time jobs, which have increased by 921K since February 2023 (from 27.020 million to 27.941 million).
Here is a summary of the labor composition in the past year: all the new jobs have been part-time jobs!
But wait there's even more, because now that the primary season is over and we enter the heart of election season and political talking points will be thrown around left and right, especially in the context of the immigration crisis created intentionally by the Biden administration which is hoping to import millions of new Democratic voters (maybe the US can hold the presidential election in Honduras or Guatemala, after all it is their citizens that will be illegally casting the key votes in November), what we find is that in February, the number of native-born workers tumbled again, sliding by a massive 560K to just 129.807 million. Add to this the December data, and we get a near-record 2.4 million plunge in native-born workers in just the past 3 months (only the covid crash was worse)!
The offset? A record 1.2 million foreign-born (read immigrants, both legal and illegal but mostly illegal) workers added in February!
Said otherwise, not only has all job creation in the past 6 years has been exclusively for foreign-born workers...

... but there has been zero job-creation for native born workers since June 2018!
This is a huge issue - especially at a time of an illegal alien flood at the southwest border...
... and is about to become a huge political scandal, because once the inevitable recession finally hits, there will be millions of furious unemployed Americans demanding a more accurate explanation for what happened - i.e., the illegal immigration floodgates that were opened by the Biden admin.
Which is also why Biden's handlers will do everything in their power to insure there is no official recession before November... and why after the election is over, all economic hell will finally break loose. Until then, however, expect the jobs numbers to get even more ridiculous.
-

 Uncategorized2 weeks ago
Uncategorized2 weeks agoAll Of The Elements Are In Place For An Economic Crisis Of Staggering Proportions
-

 Uncategorized1 month ago
Uncategorized1 month agoCathie Wood sells a major tech stock (again)
-

 Uncategorized3 weeks ago
Uncategorized3 weeks agoCalifornia Counties Could Be Forced To Pay $300 Million To Cover COVID-Era Program
-

 Uncategorized2 weeks ago
Uncategorized2 weeks agoApparel Retailer Express Moving Toward Bankruptcy
-

 Uncategorized3 weeks ago
Uncategorized3 weeks agoIndustrial Production Decreased 0.1% in January
-

 International2 days ago
International2 days agoWalmart launches clever answer to Target’s new membership program
-

 Uncategorized3 weeks ago
Uncategorized3 weeks agoRFK Jr: The Wuhan Cover-Up & The Rise Of The Biowarfare-Industrial Complex
-

 International2 days ago
International2 days agoEyePoint poaches medical chief from Apellis; Sandoz CFO, longtime BioNTech exec to retire





































