Eric Ostertag reaching the end of the line as Poseida chief, will become executive chairman; Alector exec named CEO of Pfizer-backed LNP biotech
Eric Ostertag
→ The Poseida adventure takes another turn as CEO Eric Ostertag dives into the role of executive chairman and CBO Mark Gergen steers the ship starting Feb. 1. Ostertag talked through Poseida’s early proof-of-concept data with its PSMA-target

→ The Poseida adventure takes another turn as CEO Eric Ostertag dives into the role of executive chairman and CBO Mark Gergen steers the ship starting Feb. 1. Ostertag talked through Poseida’s early proof-of-concept data with its PSMA-targeting CAR-T for prostate cancer patients in August 2021, and the San Diego biotech followed that up by inking a $3.6 billion-plus gene therapy deal with Takeda in October.
The next month, Poseida shuttered its P-BCMA-101 autologous program, with Ostertag noting that autologous CAR-T is “going the way of the dinosaur.”
Ostertag founded Poseida in 2015 and had been its lone CEO, while Gergen — who came on board in 2018 after COO stints at Halozyme and Mirati — has been president and CBO since July 2020.
 Shehnaaz Suliman
Shehnaaz Suliman→ Shehnaaz Suliman has been named CEO of ReCode Therapeutics, which is developing therapies for cystic fibrosis and primary ciliary dyskinesia using its lipid nanoparticle (LNP) delivery platform. She succeeds David Lockhart, who will continue as president but take on the role of CSO instead. A Gilead and Roche vet, Suliman is a chief executive for the first time after three years as Alector’s president and COO, and she’s also a board member at Ultragenyx and 10x Genomics. From 2017-19, Suliman was SVP, corporate development and strategy for Theravance Biopharma. ReCode has piled up the cash with a pair of $80 million financing rounds, including a Pfizer-backed Series B in October 2021.
→ While its Covid-19 pill molnupiravir grabs headlines in the advent of the Rob Davis era, Merck has named Marc Levesque VP, immunology discovery and head of the drug giant’s Exploratory Science Center in Cambridge, MA, a spokesperson tells Peer Review. Levesque hails from AbbVie, where he was medical director, immunology clinical development and later the senior director, immunology.
 David Jimenez
David Jimenez→ J&J has enlisted longtime vet David Jimenez as president of Janssen Immunology, taking over a business that includes such drugs as Tremfya and Stelara. Jimenez, who started out as a sales rep at J&J in the late 1990s, was Janssen’s president of infectious diseases from 2018-20 and had served as president of the pulmonary hypertension business since 2020.
→ Nirogy Therapeutics — co-founded by Ron DePinho and launched almost a year ago with a modest Series A to push its small molecule lactate transport inhibitor into the clinic — has tapped Simon Pedder as CEO. His predecessor, Vincent Sandanayaka, will now be Nirogy’s president and CSO. Pedder, the former head of the hepatitis franchise at Roche and the ex-CEO of Cellectar Biosciences, ended a six-year association with Athenex, where he was chief business and strategy officer.
 Riccardo Braglia
Riccardo Braglia→ Some big changes are coming Helsinn’s way and it’s starting with a CEO change. The family-owned business, run by the Braglia family, is now changing hands from Riccardo Braglia to Giorgio Calderari. Calderari has been with the company for the last 36 years and served in a variety of positions such as group general manager. COO and chief manufacturing officer. Meanwhile, Braglia will step into his dad’s old role as executive chairman.
For the rest of the family, Gabriele Braglia, founder and father to Riccardo Braglia, has been named “honorary chairman,” while his grandson, Gabriele Edoardo Braglia, joins the board of directors. Additionally, Siddharth Kaul, who hails from P&G and Novartis, is also joining the board.
→ Robert Paul is ditching his CMO title at Alector in favor of a new CEO hat at Nine Square Therapeutics — the company created in 2020 by Apple Tree Partners and UCSF scientists Matthew Jacobson, Steve Altschuler and Lani Wu. Paul started in his role as CMO of Alector back in 2016 and before that had a nearly eight-year career at Genentech in a variety of roles in the company’s medical and neuroscience clinical development organizations.
 Lucinda Crabtree
Lucinda Crabtree→ It won’t take effect until March 31, but Andrew Oakley has decided to retire as CFO of Autolus, and SVP of finance Lucinda Crabtree is all set to replace Oakley at Christian Itin’s T cell therapy shop. Oakley, the obe-cel developer’s CFO since 2018, has also been the finance chief at Actelion (from 2003-13), NovImmune, Vectura Group and Sosei Group Corporation. Crabtree, a former business analyst at AstraZeneca, first arrived at Autolus in January 2020 as head of investor relations after her time at Woodford Investment Management.
 Steven Bellon
Steven Bellon→ Carl Decicco has retired as CSO of Flagship’s Foghorn Therapeutics as Steven Bellon succeeds him at the Cigall Kadoch-founded biotech that concentrates on the chromatin regulatory system. Decicco became CSO late in 2018 after a 16-year career at Bristol Myers Squibb, where he was head of discovery. In June 2016, Bellon was selected as Foghorn’s head of discovery, and earlier at Constellation Pharmaceuticals, he served as executive director and head of structural biology, lead discovery and project management. Foghorn made some noise in the 2020 IPO boom with a $120 million offering.
 Yan Moore
Yan Moore→ Another Flagship company, Omega Therapeutics — a startup focused on epigenetic medicines that launched in September 2019 and scored $126 million from a Series C in March 2021 — has tapped Yan Moore as CMO. Moore brings Big Pharma experience from Bristol Myers, GlaxoSmithKline and Sanofi, and he was previously Ipsen’s SVP, head of oncology therapeutic area since 2018. He has also been CMO and SVP, clinical development with Anchiano, which merged with Chemomab last year.
 Matthew Klein
Matthew Klein→ Peer Review has been apprised of leadership moves at PTC Therapeutics, where Matthew Klein and Lee Golden have earned promotions. Klein, now COO, first arrived at PTC in 2019 as global head of gene & mitochondrial therapies, becoming chief development officer in 2020. Golden climbs to CMO after his stint as PTC’s head of global clinical development. Golden’s previous CMO post was with Espero BioPharma and he’s also the former SVP, therapeutic business unit head, CV, pulmonary and CNS at Mesoblast. PTC remains undaunted with its Duchenne therapy Translarna even after a Phase II fail almost a year ago.
 Martha Rook
Martha Rook→ Daphne Koller’s gang at insitro has unloaded several appointments, starting with Martha Rook (chief technical operations officer), who had held the same post at Flagship’s Sigilon and finished her 13 years at MilliporeSigma as VP, head of gene editing & novel modalities. Elsewhere, Jevan Soo Lenox (chief people officer) is a McKinsey vet who was previously the chief people and culture officer of online stylist Stitch Fix; Ellen Berg (VP, biomarker sciences) got her start as a senior scientist at AbbVie and jumps on board from Eurofins Discovery, where she was CSO of translational biology; and finally, Theofanis Karaletsos (VP, data science/machine learning) is a founding member of Uber AI Labs who had been working as a staff research scientist at Facebook, where insitro poached VP of product Tom Stocky in September.
→ The last time we saw Iovance in Peer Review, then-CEO Maria Fardis sprinted out the door as the lifileucel saga dragged on with the FDA’s request for more potency assay data in May 2021. Perhaps the appointment of an FDA lifer can expedite things as Iovance brings in Raj Puri as EVP, regulatory strategy and translational medicine sometime in Q1. Puri has 33 years of experience with the agency, 19 of those as the director of the Division of Cellular and Gene Therapies (DCGT) in the Office of Tissues and Advanced Therapies. Lifileucel keeps churning out data in the runup to a BLA submission for the tumor-infiltrating lymphocyte (TIL) therapy that’s been pushed back to this year.
 James Tursi
James Tursi→ James Tursi has been appointed EVP, global R&D of Endo, which just announced a change in marketing strategy from Pro Football Hall of Famer John Elway to everyday folks in its Dupuytren’s contracture ads. The GSK vet will finish up his time as CSO of Ferring Pharmaceuticals and start work at Endo on Jan. 18. Tursi also spent nearly two years at Antares Pharma as R&D chief and CMO, and he’s a member of the board of directors at Agile Therapeutics.
 Sohanya Cheng
Sohanya Cheng→ With first-year CEO Richard Paulson at the controls, cancer-focused Karyopharm has promoted Sohanya Cheng to chief commercial officer. Seven months ago, Karyopharm brought Cheng on board as SVP of sales and commercial operations after a short stay at Arrowhead as VP of marketing and more than a decade at Amgen. It’ll be her job to devise commercialization strategies surrounding multiple myeloma drug Xpovio (selinexor), which received a controversial rubber stamp in 2019 and an additional approval in combination with bortezomib and dexamethasone in late 2020.
 Dave Morris
Dave Morris→ A couple months after Valo Health called off its merger with SPAC partner Khosla Ventures, David Berry’s Flagship biotech has pegged Dave Morris as CMO. During his days at Novartis, Morris — who just completed a two-year run as CMO of Enterprise Therapeutics — was global head of clinical operations, analytics, and regions, and he was also operating partner of the Novartis Venture Fund. A year ago this week, Valo pulled in a $190 million Series B that ballooned to $300 million in March with additional funding from Koch Disruptive Technologies.
 Erin Colgan
Erin Colgan→ It’s not lost on Peer Review that as Season 4 of “Cobra Kai” dominates Netflix, Boston cancer player Sensei Biotherapeutics has handed out promotions to Erin Colgan and Robert Pierce. Colgan, the new CFO and a nine-year Vertex alum, had been Sensei’s SVP of finance and administration and joined the team in July 2020 from Intarcia. Pierce has made the climb to chief R&D officer after nearly two years as the CSO of John Celebi’s squad. Once the scientific director of the immunopathology lab at Fred Hutch, Pierce was the CMO/CSO at OncoSec, and during his six years at Merck, he was a medical lead for Keytruda’s trials in merkel cell carcinoma (MCC) and cutaneous T cell lymphoma (CTCL).
→ Liquid biopsy company Bluestar Genomics has pulled in David Mullarkey as CEO, taking over for acting CEO and CSO Samuel Levy (Levy will continue in his role as CSO). Mullarkey comes from another CEO stint at Omniome, where he raised over $150 million. Prior to that, Mullarkey was president and COO of Ariosa Diagnostics and held roles at J&J, Eli Lilly, Valeant Pharmaceuticals and Dow Pharmaceutical Sciences.
 Arndt Rolfs
Arndt Rolfs→ Ex-Centogene chief Arndt Rolfs has been named CEO of US-German digital healthcare and diagnostics company Arcensus as of Jan. 1, succeeding Michael Schlenk. In December 2020, Rolfs stepped down at Centogene and was replaced by Andrin Oswald, who’s been on a medical leave of absence “for a period of at least four weeks” that began just before the holidays.
 Brian Lestini
Brian Lestini→ Back in April, Peer Review told you about Brian Lestini taking the CMO job at Waltham, MA-based Pyramid Biosciences, and this week he’s worked his way up to CEO. While at Bristol Myers, Lestini had a front-row seat to such drugs as Opdivo and relatlimab as VP, oncology development before joining Pyramid Biosciences. His predecessor, co-founder Kollol Pal, will settle into the CSO post as lead candidate PBI-200 —a tropomyosin receptor kinase (TRK) inhibitor — moves through Phase I trials for such oncology targets as primary and metastatic brain cancers.
 Rosemary Harrison
Rosemary Harrison→ Rosemary Harrison has landed at T cell therapy biotech TCR² Therapeutics as chief business and strategy officer. Harrison had spent a year as SVP of corporate development and strategy at Trillium and was a key player in the $2.2 billion Pfizer buyout that was completed in November — and the drug giant wasn’t through on the M&A front, with the acquisition of Arena a month ago. From 2015-18, Harrison was head of portfolio management and strategic planning at the Novartis Institutes for Biomedical Research.
→ Sue Dillon’s Centyrin-focused Aro Biotherapeutics has brought in Sukumar Sakamuri as chief technology officer. Sakamuri, who co-founded immuno-oncology company Tollnine, was previously VP and head of chemistry at Ambrx since October 2018. Dillon and co-founder Karyn O’Neil started 2021 with an $88 million raise led by Johnson & Johnson Innovation.
 Adam Taich
Adam Taich→ Adam Taich has been named CBO of proteomics player SomaLogic, which reverse merged with CM Life Sciences II — the second SPAC from Eli Casdin and Keith Meister — back in March. Taich stepped away from Thermo Fisher Scientific after 19 years, closing out his career there as VP and general manager of the molecular biology business.
 Yvonne McGrath
Yvonne McGrath→ Immuno-oncology biotech iTeos Therapeutics has promoted Yvonne McGrath to CSO. McGrath has served as VP of R&D since June 2020, and prior to that, she was CSO at Complix. EOS-448, iTeos’ anti-TIGIT monoclonal antibody that Hal Barron is rolling the dice on at GSK in a deal worth $625 million upfront, is in early-stage trials.
 Thomas Adams
Thomas Adams→ San Diego’s Cardiff Oncology has installed Tod Smeal as CSO and Charles Monahan as SVP, regulatory affairs. Smeal changes direction here after a quick stop as chief scientist of Hexagon Bio, and the longtime Pfizer vet has also been Eli Lilly’s CSO of cancer biology. Monahan was previously the head of regulatory affairs with Erytech Pharma and has held regulatory posts at companies such as Millennium, Aveo Pharmaceuticals and Eleven Biotherapeutics.
The Cardiff Oncology family is also mourning the loss of former CEO Thomas Adams, who died Jan. 9 at age 78. Adams was chief executive when the company was known as Trovagene from June 2018 until it rebranded to Cardiff Oncology in May 2020 under current CEO Mark Erlander. From May until December 2020, Adams served as Cardiff Oncology’s executive chairman. Hepion Pharmaceuticals, where Adams had held a seat on the board since 2014, announced his death on Wednesday.
→ Gene silencing company NeuBase Therapeutics has reeled in Todd Branning as CFO. Branning joins the company after a stint as CFO at Takeda spinout Phathom Pharmaceuticals. Branning has also served as SVP, CFO at Amneal Pharmaceuticals and Teva. Earlier in his career, Branning had roles at Allergan, PricewaterhouseCoopers, PPG Industries and Merck.
 Rafal Kaminski
Rafal Kaminski→ Angelini Pharma kicked off the M&A festivities in 2021 by purchasing Arvelle Therapeutics, and the Italian biotech starts 2022 by picking up Rafal Kaminski as CSO. Kaminski spent 11 years at UCB, then left for Roche to be the Swiss pharma’s vice director, head neurosymptomatic domains, neuroscience and rare diseases. For the last two years, Kaminski led R&D and was CSO of Warsaw-based OncoArendi Therapeutics.
 Melanie Gloria
Melanie Gloria→ The crew at Los Angeles-based Acelyrin isn’t messing around as Sean Harper and Beth Seidenberg at Westlake Village Partners led a $250 million Series B in November, and the biotech has added another Horizon vet to its ranks with COO Melanie Gloria. Since 2018, Gloria had been with Tim Walbert’s team and served as SVP of development operations; prior to that, she was in clinical program development with Abbott and then AbbVie. Gloria needs no introduction to the Acelyrin C-suite: CEO Shao-Lee Lin, president Robert Carey and CMO Paul Peloso have all worked with her at Horizon.
 Ciara Kennedy
Ciara Kennedy→ Ciara Kennedy has added to her list of titles, chairing the board at Aristea Therapeutics while the San Diego inflammatory disease startup also grabs Fabio Magrini as executive medical director. Kennedy, the ex-Amplyx chief, is now the president and CEO of Sorriso Pharmaceuticals. Heading over to Aristea from Genentech, where he was principal medical director, neuroimmunology and medical director, neuroscience, Magrini’s other Big Pharma experience ranges from Pfizer to Eli Lilly.
 Star Seyedkazemi
Star Seyedkazemi→ Star Seyedkazemi has taken on the role of chief development officer at much-maligned Adverum Biotechnologies, reeling from a diabetic macular edema patient who experienced vision loss in one treated eye and mired in safety issues that further materialized in 2021. Seyedkazemi was previously VP, portfolio management for research and development at Pliant Therapeutics after serving as Allergan’s associate VP, clinical development.
 Minori Rosales
Minori Rosales→ Sesen Bio, coming off a rocky year with its bladder cancer drug Vicineum that featured a CRL and alleged serious misconduct in its 130-person study, has brought on Minori Rosales as chief development officer (effective Jan. 24) and Stephanie Vigue as director of finance (effective Jan. 17). Rosales most recently served as VP, clinical research at MacroGenics, and before that, she was a VP at MedImmune. Vigue most recently served as corporate accounting manager/interim controller at The Fi Company, and has also served as manager, corporate accounting at Spectrum Pharmaceuticals.
→ Currax Pharmaceuticals, which late last year hired Novo Nordisk vet Ed Cinca, has snagged Michael Kyle as SVP, CMO — who will lead the company’s exploration of smoking cessation opportunities through the CX-101 program. Kyle comes aboard from Jiangsu Hengrui Pharmaceuticals, where he was head of development. Kyle has also held roles at Pfizer as US medical director of established products, medical affairs; CMO, Pfizer Consumer Health; and VP, head of global clinical services and operations of Upjohn.
 Gwendolyn Binder
Gwendolyn Binder→ There are two promotions to discuss at Penn spinout Cabaletta Bio: First, Gwendolyn Binder’s title has changed from EVP to president of science and technology. She joined Cabaletta in the company’s infancy after nearly eight years as head of translational sciences and later chief technology officer of Adaptimmune. Meanwhile, Arun Das — the erstwhile executive director of new product planning and business development — moves on up to CBO.
→ Shanghai-based CAR-T player JW Therapeutics, which raised $300 million in its IPO on the Hong Kong Stock Exchange in November 2020, has announced two appointments this week: Shaun Paul Cordoba in the newly-created CSO role, and Raymond Hage as SVP of corporate development. Before joining JW, Cordoba was the executive director of synthetic biology and cell signaling at Autolus. Hage, the founder of Hapten Sciences, has also served as SVP of commercial operations and COO of Novavax.
 Stephen Smolinski
Stephen Smolinski→ With a pipeline that includes AVTX-002 for such indications as Covid-19 ARDS and inflammatory bowel syndrome, Avalo Therapeutics has locked in Stephen Smolinski as chief commercial officer. Smolinski comes to Avalo from Sobi, where was the head of US Gamifant and global immunology strategic marketing. He’s been a CCO before at Selecta Biosciences and he was head of Sanofi Genzyme’s North American rheumatology business unit at one of several stops where he gained Big Pharma commercial experience (Roche/Genentech, Bristol Myers and J&J).
→ Dominic Labriola has been appointed SVP and chief data and analytics officer at Pennsylvania-based NASH player Madrigal Pharmaceuticals. Before joining Madrigal, Labriola spent more than two decades at Bristol Myers as head of global biometric sciences.
 Philip Dana
Philip Dana→ Philip Dana has signed on as chief human resources officer of Bayer gene therapy sub AskBio, joining the likes of Jude Samulski and Kathy High on the team. Dana spent the last two years leading HR at Dendreon. After bringing AskBio into the fold, Bayer bought Vividion for $2 billion last August and struck a CRISPR deal with Mammoth just this week.
→ APIE Therapeutics is bringing on two new execs to its leadership team with the appointments of Debra Bowes as CBO and Seth Hetherington as CMO. Bowes comes aboard with stints at Chevy Chase BioPartners (CEO), Maxcyte Cell Therapy (CBO), MedImmune, Amylin Pharmaceuticals, Pfizer and Centocor. Meanwhile, Hetherington brings to the table experience from ReViral (CMO), Genocea Biosciences (CMO), Icagen (SVP of clinical and regulatory affairs), Inhibitex and GSK.
 Ana Limón
Ana Limón→ Takeda vet Ana Limón has been appointed SVP of clinical development and global medical affairs at Oryzon Genomics. Limón, who started her career at Amgen, was head of the Takeda’s oncology pipeline, global medical affairs and spent the last year as senior director and global program leader at Deciphera.
→ ARCH Ventures is adding Kaye Foster as a venture partner. Foster currently serves as a senior advisor at Boston Consulting Group. Prior to her current role, Foster was VP of global human resources at Onyx Pharmaceuticals until its acquisition by Amgen. Before that, Foster was with J&J (global human resources leader) and Pfizer.
 Ruth Krestin
Ruth Krestin→ French medtech Tissium has selected Ruth Krestin as VP of portfolio strategy. Krestin had held various roles in the last six years with AstraZeneca, including competitive intelligence director, cardiovascular, renal and metabolism (CVRM). Sofinnova led Tissium’s Series C last August that totaled $56.5 million (€50 million).
→ Pennsylvania biotech Annovis Bio — whose stock shot up 140% after seeing cognitive improvement scores increase by 30% last May for patients with either Parkinson’s disease or Alzheimer’s disease — has brought on Eve Damiano as SVP of regulatory operations. In addition, the company has promoted Cheng Fang to the position of president of R&D. Damiano joins with experience from her times at Centocor, MedImmune, OraSure Technologies and Vicuron Pharmaceuticals. Meanwhile, Fang joined Annovis last May as VP of research.
→ Stem cell-focused BioRestorative Therapies has recruited Robert Paccasassi as VP of quality assurance/regulatory compliance. Paccasassi hails from Merck KGaA, where he served as director, corporate quality systems. Before that, Paccasassi had stints at Regeneron, Millennium and Biogen.
 Paul Chu
Paul Chu→ San Diego-based Phanes Therapeutics has named Paul Chu as VP of business development. Chu joins the company from Huyabio International, where he served in the same role. Additionally, Chu has had similar roles at AiViva BioPharma, UroGen Pharma and Allergan.
→ CRO Parexel has tapped Wyatt Gotbetter as head of worldwide access consulting. Gotbetter hops aboard after a stint as a partner at Health Advances, Parexel’s independent strategic healthcare consulting unit. Before that, Gotbetter was head of new product commercialization at Biogen.
 June Lee
June Lee→ Cardio biotech CinCor Pharma, which recently hit Nasdaq with an upsized IPO after a $143 million Series B in October, has elected June Lee to the board of directors. Lee, the founder and ex-CEO of Esker Therapeutics (now Alumis), is also on the boards of Tenaya Therapeutics, Eledon Pharmaceuticals and GenEdit.
→ J&J vet Arturo Molina has been added to the board of directors at Forma Therapeutics, the Watertown, MA biotech that’s developing its lead program etavopivat for sickle cell disease. Molina has been CMO of Sutro Biopharma since 2016.
→ As Peter Thompson resigns from the board of directors at New Jersey-based PMV Pharma, the door opens for Kirsten Flowers to join a board chaired by Rich Heyman. Flowers is Kura Oncology’s chief commercial officer and the ex-SVP of commercial operations at Array Biopharma.
nasdaq covid-19 eosGovernment
The Great Replacement Loophole: Illegal Immigrants Score 5-Year Work Benefit While “Waiting” For Deporation, Asylum
The Great Replacement Loophole: Illegal Immigrants Score 5-Year Work Benefit While "Waiting" For Deporation, Asylum
Over the past several…
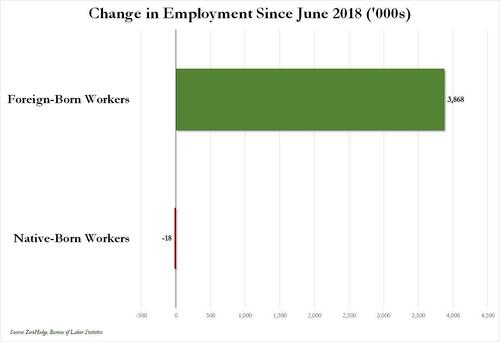
Over the past several months we've pointed out that there has been zero job creation for native-born workers since the summer of 2018...
... and that since Joe Biden was sworn into office, most of the post-pandemic job gains the administration continuously brags about have gone foreign-born (read immigrants, mostly illegal ones) workers.
And while the left might find this data almost as verboten as FBI crime statistics - as it directly supports the so-called "great replacement theory" we're not supposed to discuss - it also coincides with record numbers of illegal crossings into the United States under Biden.
In short, the Biden administration opened the floodgates, 10 million illegal immigrants poured into the country, and most of the post-pandemic "jobs recovery" went to foreign-born workers, of which illegal immigrants represent the largest chunk.

'But Tyler, illegal immigrants can't possibly work in the United States whilst awaiting their asylum hearings,' one might hear from the peanut gallery. On the contrary: ever since Biden reversed a key aspect of Trump's labor policies, all illegal immigrants - even those awaiting deportation proceedings - have been given carte blanche to work while awaiting said proceedings for up to five years...
... something which even Elon Musk was shocked to learn.
Wow, learn something new every day https://t.co/8MDtEEZGam
— Elon Musk (@elonmusk) March 10, 2024
Which leads us to another question: recall that the primary concern for the Biden admin for much of 2022 and 2023 was soaring prices, i.e., relentless inflation in general, and rising wages in particular, which in turn prompted even Goldman to admit two years ago that the diabolical wage-price spiral had been unleashed in the US (diabolical, because nothing absent a major economic shock, read recession or depression, can short-circuit it once it is in place).
Well, there is one other thing that can break the wage-price spiral loop: a flood of ultra-cheap illegal immigrant workers. But don't take our word for it: here is Fed Chair Jerome Powell himself during his February 60 Minutes interview:
PELLEY: Why was immigration important?
POWELL: Because, you know, immigrants come in, and they tend to work at a rate that is at or above that for non-immigrants. Immigrants who come to the country tend to be in the workforce at a slightly higher level than native Americans do. But that's largely because of the age difference. They tend to skew younger.
PELLEY: Why is immigration so important to the economy?
POWELL: Well, first of all, immigration policy is not the Fed's job. The immigration policy of the United States is really important and really much under discussion right now, and that's none of our business. We don't set immigration policy. We don't comment on it.
I will say, over time, though, the U.S. economy has benefited from immigration. And, frankly, just in the last, year a big part of the story of the labor market coming back into better balance is immigration returning to levels that were more typical of the pre-pandemic era.
PELLEY: The country needed the workers.
POWELL: It did. And so, that's what's been happening.
Translation: Immigrants work hard, and Americans are lazy. But much more importantly, since illegal immigrants will work for any pay, and since Biden's Department of Homeland Security, via its Citizenship and Immigration Services Agency, has made it so illegal immigrants can work in the US perfectly legally for up to 5 years (if not more), one can argue that the flood of illegals through the southern border has been the primary reason why inflation - or rather mostly wage inflation, that all too critical component of the wage-price spiral - has moderated in in the past year, when the US labor market suddenly found itself flooded with millions of perfectly eligible workers, who just also happen to be illegal immigrants and thus have zero wage bargaining options.
None of this is to suggest that the relentless flood of immigrants into the US is not also driven by voting and census concerns - something Elon Musk has been pounding the table on in recent weeks, and has gone so far to call it "the biggest corruption of American democracy in the 21st century", but in retrospect, one can also argue that the only modest success the Biden admin has had in the past year - namely bringing inflation down from a torrid 9% annual rate to "only" 3% - has also been due to the millions of illegals he's imported into the country.
We would be remiss if we didn't also note that this so often carries catastrophic short-term consequences for the social fabric of the country (the Laken Riley fiasco being only the latest example), not to mention the far more dire long-term consequences for the future of the US - chief among them the trillions of dollars in debt the US will need to incur to pay for all those new illegal immigrants Democrat voters and low-paid workers. This is on top of the labor revolution that will kick in once AI leads to mass layoffs among high-paying, white-collar jobs, after which all those newly laid off native-born workers hoping to trade down to lower paying (if available) jobs will discover that hardened criminals from Honduras or Guatemala have already taken them, all thanks to Joe Biden.
Spread & Containment
‘I couldn’t stand the pain’: the Turkish holiday resort that’s become an emergency dental centre for Britons who can’t get treated at home
The crisis in NHS dentistry is driving increasing numbers abroad for treatment. Here are some of their stories.

It’s a hot summer day in the Turkish city of Antalya, a Mediterranean resort with golden beaches, deep blue sea and vibrant nightlife. The pool area of the all-inclusive resort is crammed with British people on sun loungers – but they aren’t here for a holiday. This hotel is linked to a dental clinic that organises treatment packages, and most of these guests are here to see a dentist.
From Norwich, two women talk about gums and injections. A man from Wales holds a tissue close to his mouth and spits blood – he has just had two molars extracted.
The dental clinic organises everything for these dental “tourists” throughout their treatment, which typically lasts from three to 15 days. The stories I hear of what has caused them to travel to Turkey are strikingly similar: all have struggled to secure dental treatment at home on the NHS.
“The hotel is nice and some days I go to the beach,” says Susan*, a hairdresser in her mid-30s from Norwich. “But really, we aren’t tourists like in a proper holiday. We come here because we have no choice. I couldn’t stand the pain.”

This is Susan’s second visit to Antalya. She explains that her ordeal started two years earlier:
I went to an NHS dentist who told me I had gum disease … She did some cleaning to my teeth and gums but it got worse. When I ate, my teeth were moving … the gums were bleeding and it was very painful. I called to say I was in pain but the clinic was not accepting NHS patients any more.
The only option the dentist offered Susan was to register as a private patient:
I asked how much. They said £50 for x-rays and then if the gum disease got worse, £300 or so for extraction. Four of them were moving – imagine: £1,200 for losing your teeth! Without teeth I’d lose my clients, but I didn’t have the money. I’m a single mum. I called my mum and cried.
Susan’s mother told her about a friend of hers who had been to Turkey for treatment, then together they found a suitable clinic:
The prices are so much cheaper! Tooth extraction, x-rays, consultations – it all comes included. The flight and hotel for seven days cost the same as losing four teeth in Norwich … I had my lower teeth removed here six months ago, now I’ve got implants … £2,800 for everything – hotel, transfer, treatments. I only paid the flights separately.
In the UK, roughly half the adult population suffers from periodontitis – inflammation of the gums caused by plaque bacteria that can lead to irreversible loss of gums, teeth, and bone. Regular reviews by a dentist or hygienist are required to manage this condition. But nine out of ten dental practices cannot offer NHS appointments to new adult patients, while eight in ten are not accepting new child patients.
Some UK dentists argue that Britons who travel abroad for treatment do so mainly for cosmetic procedures. They warn that dental tourism is dangerous, and that if their treatment goes wrong, dentists in the UK will be unable to help because they don’t want to be responsible for further damage. Susan shrugs this off:
Dentists in England say: ‘If you go to Turkey, we won’t touch you [afterwards].’ But I don’t worry because there are no appointments at home anyway. They couldn’t help in the first place, and this is why we are in Turkey.
‘How can we pay all this money?’
As a social anthropologist, I travelled to Turkey a number of times in 2023 to investigate the crisis of NHS dentistry, and the journeys abroad that UK patients are increasingly making as a result. I have relatives in Istanbul and have been researching migration and trading patterns in Turkey’s largest city since 2016.
In August 2023, I visited the resort in Antalya, nearly 400 miles south of Istanbul. As well as Susan, I met a group from a village in Wales who said there was no provision of NHS dentistry back home. They had organised a two-week trip to Turkey: the 12-strong group included a middle-aged couple with two sons in their early 20s, and two couples who were pensioners. By going together, Anya tells me, they could support each other through their different treatments:
I’ve had many cavities since I was little … Before, you could see a dentist regularly – you didn’t even think about it. If you had pain or wanted a regular visit, you phoned and you went … That was in the 1990s, when I went to the dentist maybe every year.
Anya says that once she had children, her family and work commitments meant she had no time to go to the dentist. Then, years later, she started having serious toothache:
Every time I chewed something, it hurt. I ate soups and soft food, and I also lost weight … Even drinking was painful – tea: pain, cold water: pain. I was taking paracetamol all the time! I went to the dentist to fix all this, but there were no appointments.
Anya was told she would have to wait months, or find a dentist elsewhere:
A private clinic gave me a list of things I needed done. Oh my God, almost £6,000. My husband went too – same story. How can we pay all this money? So we decided to come to Turkey. Some people we know had been here, and others in the village wanted to come too. We’ve brought our sons too – they also need to be checked and fixed. Our whole family could be fixed for less than £6,000.
By the time they travelled, Anya’s dental problems had turned into a dental emergency. She says she could not live with the pain anymore, and was relying on paracetamol.
In 2023, about 6 million adults in the UK experienced protracted pain (lasting more than two weeks) caused by toothache. Unintentional paracetamol overdose due to dental pain is a significant cause of admissions to acute medical units. If left untreated, tooth infections can spread to other parts of the body and cause life-threatening complications – and on rare occasions, death.
In February 2024, police were called to manage hundreds of people queuing outside a newly opened dental clinic in Bristol, all hoping to be registered or seen by an NHS dentist. One in ten Britons have admitted to performing “DIY dentistry”, of which 20% did so because they could not find a timely appointment. This includes people pulling out their teeth with pliers and using superglue to repair their teeth.
In the 1990s, dentistry was almost entirely provided through NHS services, with only around 500 solely private dentists registered. Today, NHS dentist numbers in England are at their lowest level in a decade, with 23,577 dentists registered to perform NHS work in 2022-23, down 695 on the previous year. Furthermore, the precise division of NHS and private work that each dentist provides is not measured.
The COVID pandemic created longer waiting lists for NHS treatment in an already stretched public service. In Bridlington, Yorkshire, people are now reportedly having to wait eight-to-nine years to get an NHS dental appointment with the only remaining NHS dentist in the town.
In his book Patients of the State (2012), Argentine sociologist Javier Auyero describes the “indignities of waiting”. It is the poor who are mostly forced to wait, he writes. Queues for state benefits and public services constitute a tangible form of power over the marginalised. There is an ethnic dimension to this story, too. Data suggests that in the UK, patients less likely to be effective in booking an NHS dental appointment are non-white ethnic groups and Gypsy or Irish travellers, and that it is particularly challenging for refugees and asylum-seekers to access dental care.

This article is part of Conversation Insights
The Insights team generates long-form journalism derived from interdisciplinary research. The team is working with academics from different backgrounds who have been engaged in projects aimed at tackling societal and scientific challenges.
In 2022, I experienced my own dental emergency. An infected tooth was causing me debilitating pain, and needed root canal treatment. I was advised this would cost £71 on the NHS, plus £307 for a follow-up crown – but that I would have to wait months for an appointment. The pain became excruciating – I could not sleep, let alone wait for months. In the same clinic, privately, I was quoted £1,300 for the treatment (more than half my monthly income at the time), or £295 for a tooth extraction.
I did not want to lose my tooth because of lack of money. So I bought a flight to Istanbul immediately for the price of the extraction in the UK, and my tooth was treated with root canal therapy by a private dentist there for £80. Including the costs of travelling, the total was a third of what I was quoted to be treated privately in the UK. Two years on, my treated tooth hasn’t given me any more problems.
A better quality of life
Not everyone is in Antalya for emergency procedures. The pensioners from Wales had contacted numerous clinics they found on the internet, comparing prices, treatments and hotel packages at least a year in advance, in a carefully planned trip to get dental implants – artificial replacements for tooth roots that help support dentures, crowns and bridges.

In Turkey, all the dentists I speak to (most of whom cater mainly for foreigners, including UK nationals) consider implants not a cosmetic or luxurious treatment, but a development in dentistry that gives patients who are able to have the procedure a much better quality of life. This procedure is not available on the NHS for most of the UK population, and the patients I meet in Turkey could not afford implants in private clinics back home.
Paul is in Antalya to replace his dentures, which have become uncomfortable and irritating to his gums, with implants. He says he couldn’t find an appointment to see an NHS dentist. His wife Sonia went through a similar procedure the year before and is very satisfied with the results, telling me: “Why have dentures that you need to put in a glass overnight, in the old style? If you can have implants, I say, you’re better off having them.”
Most of the dental tourists I meet in Antalya are white British: this city, known as the Turkish Riviera, has developed an entire economy catering to English-speaking tourists. In 2023, more than 1.3 million people visited the city from the UK, up almost 15% on the previous year.
Read more: NHS dentistry is in crisis – are overseas dentists the answer?
In contrast, the Britons I meet in Istanbul are predominantly from a non-white ethnic background. Omar, a pensioner of Pakistani origin in his early 70s, has come here after waiting “half a year” for an NHS appointment to fix the dental bridge that is causing him pain. Omar’s son had been previously for a hair transplant, and was offered a free dental checkup by the same clinic, so he suggested it to his father. Having worked as a driver for a manufacturing company for two decades in Birmingham, Omar says he feels disappointed to have contributed to the British economy for so long, only to be “let down” by the NHS:
At home, I must wait and wait and wait to get a bridge – and then I had many problems with it. I couldn’t eat because the bridge was uncomfortable and I was in pain, but there were no appointments on the NHS. I asked a private dentist and they recommended implants, but they are far too expensive [in the UK]. I started losing weight, which is not a bad thing at the beginning, but then I was worrying because I couldn’t chew and eat well and was losing more weight … Here in Istanbul, I got dental implants – US$500 each, problem solved! In England, each implant is maybe £2,000 or £3,000.
In the waiting area of another clinic in Istanbul, I meet Mariam, a British woman of Iraqi background in her late 40s, who is making her second visit to the dentist here. Initially, she needed root canal therapy after experiencing severe pain for weeks. Having been quoted £1,200 in a private clinic in outer London, Mariam decided to fly to Istanbul instead, where she was quoted £150 by a dentist she knew through her large family. Even considering the cost of the flight, Mariam says the decision was obvious:
Dentists in England are so expensive and NHS appointments so difficult to find. It’s awful there, isn’t it? Dentists there blamed me for my rotten teeth. They say it’s my fault: I don’t clean or I ate sugar, or this or that. I grew up in a village in Iraq and didn’t go to the dentist – we were very poor. Then we left because of war, so we didn’t go to a dentist … When I arrived in London more than 20 years ago, I didn’t speak English, so I still didn’t go to the dentist … I think when you move from one place to another, you don’t go to the dentist unless you are in real, real pain.
In Istanbul, Mariam has opted not only for the urgent root canal treatment but also a longer and more complex treatment suggested by her consultant, who she says is a renowned doctor from Syria. This will include several extractions and implants of back and front teeth, and when I ask what she thinks of achieving a “Hollywood smile”, Mariam says:
Who doesn’t want a nice smile? I didn’t come here to be a model. I came because I was in pain, but I know this doctor is the best for implants, and my front teeth were rotten anyway.
Dentists in the UK warn about the risks of “overtreatment” abroad, but Mariam appears confident that this is her opportunity to solve all her oral health problems. Two of her sisters have already been through a similar treatment, so they all trust this doctor.

The UK’s ‘dental deserts’
To get a fuller understanding of the NHS dental crisis, I’ve also conducted 20 interviews in the UK with people who have travelled or were considering travelling abroad for dental treatment.
Joan, a 50-year-old woman from Exeter, tells me she considered going to Turkey and could have afforded it, but that her back and knee problems meant she could not brave the trip. She has lost all her lower front teeth due to gum disease and, when I meet her, has been waiting 13 months for an NHS dental appointment. Joan tells me she is living in “shame”, unable to smile.
In the UK, areas with extremely limited provision of NHS dental services – known as as “dental deserts” – include densely populated urban areas such as Portsmouth and Greater Manchester, as well as many rural and coastal areas.
In Felixstowe, the last dentist taking NHS patients went private in 2023, despite the efforts of the activist group Toothless in Suffolk to secure better access to NHS dentists in the area. It’s a similar story in Ripon, Yorkshire, and in Dumfries & Galloway, Scotland, where nearly 25,000 patients have been de-registered from NHS dentists since 2021.
Data shows that 2 million adults must travel at least 40 miles within the UK to access dental care. Branding travel for dental care as “tourism” carries the risk of disguising the elements of duress under which patients move to restore their oral health – nationally and internationally. It also hides the immobility of those who cannot undertake such journeys.
The 90-year-old woman in Dumfries & Galloway who now faces travelling for hours by bus to see an NHS dentist can hardly be considered “tourism” – nor the Ukrainian war refugees who travelled back from West Sussex and Norwich to Ukraine, rather than face the long wait to see an NHS dentist.
Many people I have spoken to cannot afford the cost of transport to attend dental appointments two hours away – or they have care responsibilities that make it impossible. Instead, they are forced to wait in pain, in the hope of one day securing an appointment closer to home.

‘Your crisis is our business’
The indignities of waiting in the UK are having a big impact on the lives of some local and foreign dentists in Turkey. Some neighbourhoods are rapidly changing as dental and other health clinics, usually in luxurious multi-storey glass buildings, mushroom. In the office of one large Istanbul medical complex with sections for hair transplants and dentistry (plus one linked to a hospital for more extensive cosmetic surgery), its Turkish owner and main investor tells me:
Your crisis is our business, but this is a bazaar. There are good clinics and bad clinics, and unfortunately sometimes foreign patients do not know which one to choose. But for us, the business is very good.
This clinic only caters to foreign patients. The owner, an architect by profession who also developed medical clinics in Brazil, describes how COVID had a major impact on his business:
When in Europe you had COVID lockdowns, Turkey allowed foreigners to come. Many people came for ‘medical tourism’ – we had many patients for cosmetic surgery and hair transplants. And that was when the dental business started, because our patients couldn’t see a dentist in Germany or England. Then more and more patients started to come for dental treatments, especially from the UK and Ireland. For them, it’s very, very cheap here.
The reasons include the value of the Turkish lira relative to the British pound, the low cost of labour, the increasing competition among Turkish clinics, and the sheer motivation of dentists here. While most dentists catering to foreign patients are from Turkey, others have arrived seeking refuge from war and violence in Syria, Iraq, Afghanistan, Iran and beyond. They work diligently to rebuild their lives, careers and lost wealth.
Regardless of their origin, all dentists in Turkey must be registered and certified. Hamed, a Syrian dentist and co-owner of a new clinic in Istanbul catering to European and North American patients, tells me:
I know that you say ‘Syrian’ and people think ‘migrant’, ‘refugee’, and maybe think ‘how can this dentist be good?’ – but Syria, before the war, had very good doctors and dentists. Many of us came to Turkey and now I have a Turkish passport. I had to pass the exams to practise dentistry here – I study hard. The exams are in Turkish and they are difficult, so you cannot say that Syrian doctors are stupid.
Hamed talks excitedly about the latest technology that is coming to his profession: “There are always new materials and techniques, and we cannot stop learning.” He is about to travel to Paris to an international conference:
I can say my techniques are very advanced … I bet I put more implants and do more bone grafting and surgeries every week than any dentist you know in England. A good dentist is about practice and hand skills and experience. I work hard, very hard, because more and more patients are arriving to my clinic, because in England they don’t find dentists.

While there is no official data about the number of people travelling from the UK to Turkey for dental treatment, investors and dentists I speak to consider that numbers are rocketing. From all over the world, Turkey received 1.2 million visitors for “medical tourism” in 2022, an increase of 308% on the previous year. Of these, about 250,000 patients went for dentistry. One of the most renowned dental clinics in Istanbul had only 15 British patients in 2019, but that number increased to 2,200 in 2023 and is expected to reach 5,500 in 2024.
Like all forms of medical care, dental treatments carry risks. Most clinics in Turkey offer a ten-year guarantee for treatments and a printed clinical history of procedures carried out, so patients can show this to their local dentists and continue their regular annual care in the UK. Dental treatments, checkups and maintaining a good oral health is a life-time process, not a one-off event.
Many UK patients, however, are caught between a rock and a hard place – criticised for going abroad, yet unable to get affordable dental care in the UK before and after their return. The British Dental Association has called for more action to inform these patients about the risks of getting treated overseas – and has warned UK dentists about the legal implications of treating these patients on their return. But this does not address the difficulties faced by British patients who are being forced to go abroad in search of affordable, often urgent dental care.
A global emergency
The World Health Organization states that the explosion of oral disease around the world is a result of the “negligent attitude” that governments, policymakers and insurance companies have towards including oral healthcare under the umbrella of universal healthcare. It as if the health of our teeth and mouth is optional; somehow less important than treatment to the rest of our body. Yet complications from untreated tooth decay can lead to hospitalisation.
The main causes of oral health diseases are untreated tooth decay, severe gum disease, toothlessness, and cancers of the lip and oral cavity. Cases grew during the pandemic, when little or no attention was paid to oral health. Meanwhile, the global cosmetic dentistry market is predicted to continue growing at an annual rate of 13% for the rest of this decade, confirming the strong relationship between socioeconomic status and access to oral healthcare.
In the UK since 2018, there have been more than 218,000 admissions to hospital for rotting teeth, of which more than 100,000 were children. Some 40% of children in the UK have not seen a dentist in the past 12 months. The role of dentists in prevention of tooth decay and its complications, and in the early detection of mouth cancer, is vital. While there is a 90% survival rate for mouth cancer if spotted early, the lack of access to dental appointments is causing cases to go undetected.
The reasons for the crisis in NHS dentistry are complex, but include: the real-term cuts in funding to NHS dentistry; the challenges of recruitment and retention of dentists in rural and coastal areas; pay inequalities facing dental nurses, most of them women, who are being badly hit by the cost of living crisis; and, in England, the 2006 Dental Contract that does not remunerate dentists in a way that encourages them to continue seeing NHS patients.
The UK is suffering a mass exodus of the public dentistry workforce, with workers leaving the profession entirely or shifting to the private sector, where payments and life-work balance are better, bureaucracy is reduced, and prospects for career development look much better. A survey of general dental practitioners found that around half have reduced their NHS work since the pandemic – with 43% saying they were likely to go fully private, and 42% considering a career change or taking early retirement.
Reversing the UK’s dental crisis requires more commitment to substantial reform and funding than the “recovery plan” announced by Victoria Atkins, the secretary of state for health and social care, on February 7.
The stories I have gathered show that people travelling abroad for dental treatment don’t see themselves as “tourists” or vanity-driven consumers of the “Hollywood smile”. Rather, they have been forced by the crisis in NHS dentistry to seek out a service 1,500 miles away in Turkey that should be a basic, affordable right for all, on their own doorstep.
*Names in this article have been changed to protect the anonymity of the interviewees.

For you: more from our Insights series:
GP crisis: how did things go so wrong, and what needs to change?
Insomnia: how chronic sleep problems can lead to a spiralling decline in mental health
To hear about new Insights articles, join the hundreds of thousands of people who value The Conversation’s evidence-based news. Subscribe to our newsletter.
Diana Ibanez Tirado receives funding from the School of Global Studies, University of Sussex.
pound pandemic treatment therapy spread recovery iran brazil european europe uk germany ukraine world health organizationInternational
Beloved mall retailer files Chapter 7 bankruptcy, will liquidate
The struggling chain has given up the fight and will close hundreds of stores around the world.

It has been a brutal period for several popular retailers. The fallout from the covid pandemic and a challenging economic environment have pushed numerous chains into bankruptcy with Tuesday Morning, Christmas Tree Shops, and Bed Bath & Beyond all moving from Chapter 11 to Chapter 7 bankruptcy liquidation.
In all three of those cases, the companies faced clear financial pressures that led to inventory problems and vendors demanding faster, or even upfront payment. That creates a sort of inevitability.
Related: Beloved retailer finds life after bankruptcy, new famous owner
When a retailer faces financial pressure it sets off a cycle where vendors become wary of selling them items. That leads to barren shelves and no ability for the chain to sell its way out of its financial problems.
Once that happens bankruptcy generally becomes the only option. Sometimes that means a Chapter 11 filing which gives the company a chance to negotiate with its creditors. In some cases, deals can be worked out where vendors extend longer terms or even forgive some debts, and banks offer an extension of loan terms.
In other cases, new funding can be secured which assuages vendor concerns or the company might be taken over by its vendors. Sometimes, as was the case with David's Bridal, a new owner steps in, adds new money, and makes deals with creditors in order to give the company a new lease on life.
It's rare that a retailer moves directly into Chapter 7 bankruptcy and decides to liquidate without trying to find a new source of funding.
Image source: Getty Images
The Body Shop has bad news for customers
The Body Shop has been in a very public fight for survival. Fears began when the company closed half of its locations in the United Kingdom. That was followed by a bankruptcy-style filing in Canada and an abrupt closure of its U.S. stores on March 4.
"The Canadian subsidiary of the global beauty and cosmetics brand announced it has started restructuring proceedings by filing a Notice of Intention (NOI) to Make a Proposal pursuant to the Bankruptcy and Insolvency Act (Canada). In the same release, the company said that, as of March 1, 2024, The Body Shop US Limited has ceased operations," Chain Store Age reported.
A message on the company's U.S. website shared a simple message that does not appear to be the entire story.
"We're currently undergoing planned maintenance, but don't worry we're due to be back online soon."
That same message is still on the company's website, but a new filing makes it clear that the site is not down for maintenance, it's down for good.
The Body Shop files for Chapter 7 bankruptcy
While the future appeared bleak for The Body Shop, fans of the brand held out hope that a savior would step in. That's not going to be the case.
The Body Shop filed for Chapter 7 bankruptcy in the United States.
"The US arm of the ethical cosmetics group has ceased trading at its 50 outlets. On Saturday (March 9), it filed for Chapter 7 insolvency, under which assets are sold off to clear debts, putting about 400 jobs at risk including those in a distribution center that still holds millions of dollars worth of stock," The Guardian reported.
After its closure in the United States, the survival of the brand remains very much in doubt. About half of the chain's stores in the United Kingdom remain open along with its Australian stores.
The future of those stores remains very much in doubt and the chain has shared that it needs new funding in order for them to continue operating.
The Body Shop did not respond to a request for comment from TheStreet.
bankruptcy pandemic canada-

 Uncategorized2 weeks ago
Uncategorized2 weeks agoAll Of The Elements Are In Place For An Economic Crisis Of Staggering Proportions
-

 Uncategorized1 month ago
Uncategorized1 month agoCathie Wood sells a major tech stock (again)
-

 Uncategorized3 weeks ago
Uncategorized3 weeks agoCalifornia Counties Could Be Forced To Pay $300 Million To Cover COVID-Era Program
-

 Uncategorized2 weeks ago
Uncategorized2 weeks agoApparel Retailer Express Moving Toward Bankruptcy
-

 Uncategorized3 weeks ago
Uncategorized3 weeks agoIndustrial Production Decreased 0.1% in January
-

 International2 days ago
International2 days agoWalmart launches clever answer to Target’s new membership program
-

 International3 days ago
International3 days agoEyePoint poaches medical chief from Apellis; Sandoz CFO, longtime BioNTech exec to retire
-

 Uncategorized3 weeks ago
Uncategorized3 weeks agoRFK Jr: The Wuhan Cover-Up & The Rise Of The Biowarfare-Industrial Complex























